
The reduction of antibiotic growth promoters in swine and poultry production has led nutritionists to rely on a wide range of non-antibiotic antibacterial ingredients. Organic acids, zinc oxide, botanicals, fatty acids and biopolymers are now used routinely to manage gut bacterial pressure. However, these ingredients do not work in the same way. Their efficacy depends on whether they act in a bacteriostatic or bactericidal manner, and on how they are used within a broader feeding strategy.
Understanding these differences is essential to design effective, consistent and economical gut health programs.
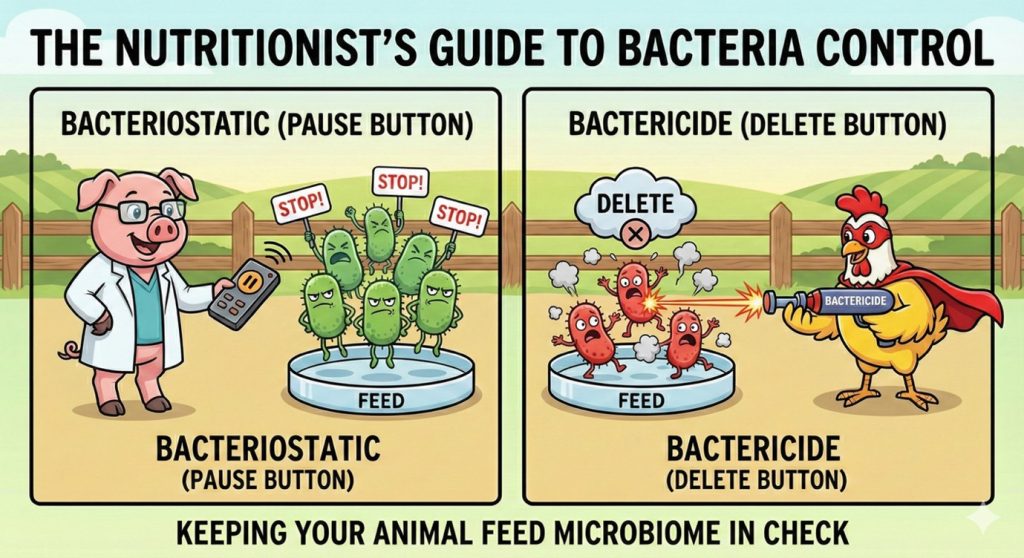
Bacteriostatic versus bactericidal actions
Bacteriostatic ingredients slow down bacterial growth and replication without killing bacteria directly. By limiting multiplication, they reduce the speed at which bacterial populations expand and allow the animal and its commensal microbiota to maintain control. Their efficacy depends primarily on time of exposure. They must be present continuously to exert meaningful pressure.
In a typical bacteriostatic mechanism, the active molecule penetrates the bacterial cell in a neutral form and dissociates inside the cytoplasm. This intracellular dissociation disturbs metabolic balance and forces the bacterium to activate energy-consuming systems to restore internal stability. ATP is redirected from growth toward survival. As energy reserves decline, protein synthesis slows, DNA replication is delayed, and cell division is postponed.
The cell remains structurally intact, but its capacity to multiply is reduced. As long as exposure continues, proliferation remains limited. Once exposure stops, surviving bacteria may resume growth. This example illustrates the core principle of bacteriostatic action: metabolic restraint over time rather than structural destruction.
Beyond this metabolic ATP-stress model, bacteriostatic effects can also arise from inhibition of protein synthesis, interference with DNA replication, disruption of quorum sensing and bacterial communication systems, limitation of nutrient availability, or binding of key bacterial enzymes. In all these cases, the defining characteristic remains the same: bacterial growth is slowed without immediate cell death.
While bacteriostatic mechanisms focus on slowing or restraining bacterial expansion, bactericidal mechanisms shift the objective toward eliminating bacterial cells through irreversible damage.
In a typical bactericidal mechanism, the active compound interacts directly with the bacterial membrane. Upon contact, it destabilizes the lipid bilayer that maintains cellular integrity. Membrane permeability increases, ion gradients collapse, and essential intracellular components leak into the surrounding environment. Once structural disruption exceeds a critical threshold, the damage becomes irreversible and the bacterium dies.
Here, the outcome depends primarily on reaching a sufficient local concentration to compromise membrane integrity. Below that concentration, the cell may recover. Above it, survival is no longer possible. This example illustrates the core principle of bactericidal action: irreversible damage once a lethal threshold is reached.
While bactericidal mechanisms may also involve oxidative damage, direct DNA injury, enzyme inactivation, or metal-induced cellular toxicity, they all share a common endpoint: structural or molecular damage that the bacterial cell cannot repair.
Despite the diversity of molecular targets, the fundamental distinction remains consistent: bacteriostatic actions limit replication and depend on exposure time, whereas bactericidal actions induce irreversible cellular damage and depend on achieving sufficient local concentration.
Bacteriostatic ingredients
Short-chain organic acids (SCFA)
Formic, lactic, citric, fumaric and propionic acids act mainly through intracellular acidification, disrupting enzymatic activity and energy metabolism and slowing bacterial replication. They are particularly effective against gram-negative bacteria.
Their value lies in early and continuous exposure, not in peak concentrations. Used correctly, they reduce the speed of bacterial proliferation and lower downstream bacterial pressure.
Aromatic organic acids
Benzoic and sorbic acids remain active further down the gut at higher pH because of their higher pKa, which maintains a significant fraction of the molecules in the undissociated form even when pH increases. As a result, their bacteriostatic effect relies less on rapid intracellular acidification, as seen with short-chain organic acids, and more on sustained interference with bacterial energy metabolism and enzymatic pathways. This makes them particularly relevant for intestinal and hindgut stabilization.
Plant extracts and essential oils
At very low inclusion levels, many botanical compounds interfere with bacterial metabolism, enzyme systems and cell-to-cell communication rather than killing cells outright. That groups contains products like cinnamaldehyde, carvacrol, thymol, tannins, catechins, garlic extract.
Their effect is mostly bacteriostatic and depends on continuous exposure rather than high dosage.
Bactericidal ingredients
Short-chain organic acids (SCFA)
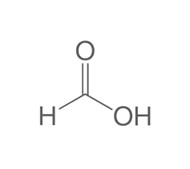
Formic, lactic, citric, fumaric and propionic acids act mainly through intracellular acidification, disrupting enzymatic activity and energy metabolism and slowing bacterial replication. They are particularly effective against gram-negative bacteria.
Their value lies in early and continuous exposure, not in peak concentrations. Used correctly, they reduce the speed of bacterial proliferation and lower downstream bacterial pressure.
Aromatic organic acids
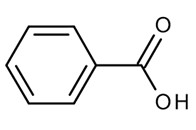
Benzoic and sorbic acids remain active further down the gut at higher pH because of their higher pKa, which maintains a significant fraction of the molecules in the undissociated form even when pH increases. As a result, their bacteriostatic effect relies less on rapid intracellular acidification, as seen with short-chain organic acids, and more on sustained interference with bacterial energy metabolism and enzymatic pathways. This makes them particularly relevant for intestinal and hindgut stabilization.
Plant extracts and essential oils
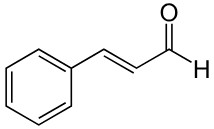
At very low inclusion levels, many botanical compounds interfere with bacterial metabolism, enzyme systems and cell-to-cell communication rather than killing cells outright. That groups contains products like cinnamaldehyde, carvacrol, thymol, tannins, catechins, garlic extract.
Their effect is mostly bacteriostatic and depends on continuous exposure rather than high dosage.
Bactericidal ingredients
Zinc oxide and Copper

Copper and zinc ions exert antibacterial effects through direct interactions with bacterial membranes and proteins. Copper (both oxide and sulfate and Zinc (only oxide) have an additional and important mode of action through the generation of reactive oxygen species (ROS – highly reactive free radicals) at the bacterial surface, leading to oxidative damage and cell death.
The antibacterial efficacy of zinc oxide is highly dose dependent. At nutritional levels, the effect is limited. At elevated or pharmacological levels, bactericidal activity increases sharply. Because the margin between inefficacy and excess can be narrow, copper and zinc must be considered precision bactericidal tools rather than background stabilizers.
Medium-chain fatty acids
Caprylic, capric and lauric acids are effective bactericides because of their amphiphilic nature. Their molecules contain both hydrophilic and lipophilic domains, allowing them to insert directly into bacterial cell membranes.
This amphiphilic property disrupts membrane integrity, causes leakage of intracellular content and leads to rapid bacterial death. Medium-chain fatty acids are particularly effective against gram-positive bacteria and require accurate dosing to be effective.
Phenolic essential oils
At higher inclusion levels, compounds such as thymol and carvacrol become bactericidal. They destabilize bacterial membranes and proteins, leading to irreversible cell damage. Their use requires careful formulation to balance efficacy and tolerance.
Chitosan and polycationic biopolymers
Chitosan is a positively charged biopolymer that interacts directly with negatively charged bacterial surfaces. This electrostatic interaction increases membrane permeability and reduces bacterial viability, with a strong preference for gram-negative bacteria such as e. coli and salmonella.
At lower inclusion, chitosan mainly limits growth and adhesion. At higher functional levels, it behaves as a bactericidal agent. Its efficacy depends on dose and dispersion rather than exposure time.
In non-antibiotic gut health strategies, the most robust programs do not rely on single ingredients, but on deliberate synergies between bacteriostatic and bactericidal tools.
Bacteriostatic ingredients act over time, limiting proliferation and progressively stressing bacterial physiology. Bactericidal ingredients act through dose-dependent killing. When combined, bacteriostatic pressure prepares bacteria to be killed more efficiently.
Two combinations illustrate this principle particularly well.
Formic acid and zinc oxide
Formic acid provides continuous bacteriostatic stress by slowing bacterial replication and disturbing metabolic activity from the earliest stages of digestion. Under sustained acid pressure, bacteria must constantly expend energy to maintain intracellular pH and basic cellular functions, resulting in metabolic stress and reduced resilience.
Zinc oxide is a dose-dependent bactericidal tool whose efficacy increases when bacteria are already weakened. Acid-stressed bacteria show greater membrane fragility and impaired protein systems, making zinc-mediated killing more efficient at a given dose.
This synergy is particularly effective in the stomach, where low pH maximizes the activity of short-chain organic acids and increases zinc oxide solubility, improving zinc ion availability. In this environment, formic acid weakens bacterial defenses while zinc oxide delivers the decisive bactericidal effect.
Benzoic acid and chitosan
Benzoic acid provides sustained bacteriostatic pressure deeper in the gut, progressively weakening bacterial metabolism over time. Its action limits proliferation and reduces bacterial resilience without relying on strong acidification.
Chitosan is a contact-active bactericidal tool whose efficacy depends on direct interaction with bacterial surfaces. When bacteria are metabolically constrained by benzoic acid, their ability to maintain membrane integrity and repair damage is reduced, making chitosan more effective.
This combination delivers a durable reduction in bacterial pressure while avoiding aggressive dosing, and is particularly relevant for intestinal stability or when zinc oxide use is limited.
Conclusion: Strategic learnings for nutritionists
Three key learnings emerge from understanding bacteriostatic and bactericidal modes of action.
First, bacteriostatic efficacy is driven by time of exposure, while bactericidal efficacy is driven by dosage. Confusing these two leads to inconsistent field results.
Second, bacteriostatic strategies are most effective when they act early, before bacterial populations expand, reducing downstream pressure and improving overall control. Third, robust gut health programs are built on synergy, not on single ingredients. Associating bacteriostatic and bactericidal tools improves consistency, reduces reliance on extreme dosages, and forms the foundation of effective agp-free strategies adapted to swine and poultry production in Vietnam and Thailand.
David Serene
Nutrispices Director





